The Limits of Conventional Cancer Treatments
Cancer has been with us since before Homo Sapiens was a species distinct from our predecessors. Indeed, it plagued fish hundreds of millions of years before primates evolved [1]. The first written record of its diagnosis and treatment over four millennia ago involved incising hard tumors that had protruded from the body [2]. The examination, diagnosis, and treatment of cancer have accompanied humanity through all our existence. Medical science has now reached a point where some cancers are considered curable, but many treatments involve significant risks. Some of the most significant side effects occur because treatments are non-specific. Common, nonsurgical treatments like radiation and chemotherapy work by damaging all cells. The logic is sound—expose ever hungry, ever-growing tumors to something so toxic they could never recover. However, the technology is flawed because the treatments cannot discriminate between healthy and cancerous cells. As a result, damage can occur throughout the body, particularly in rapidly dividing tissues such as bone marrow, the digestive tract, and hair follicles [3]. In the best case, these treatments are agonizing but ultimately successful and do not cause lasting damage. In the worst, they cause life-threatening harm, and patients die from a combination of damage from cancer and its cure.
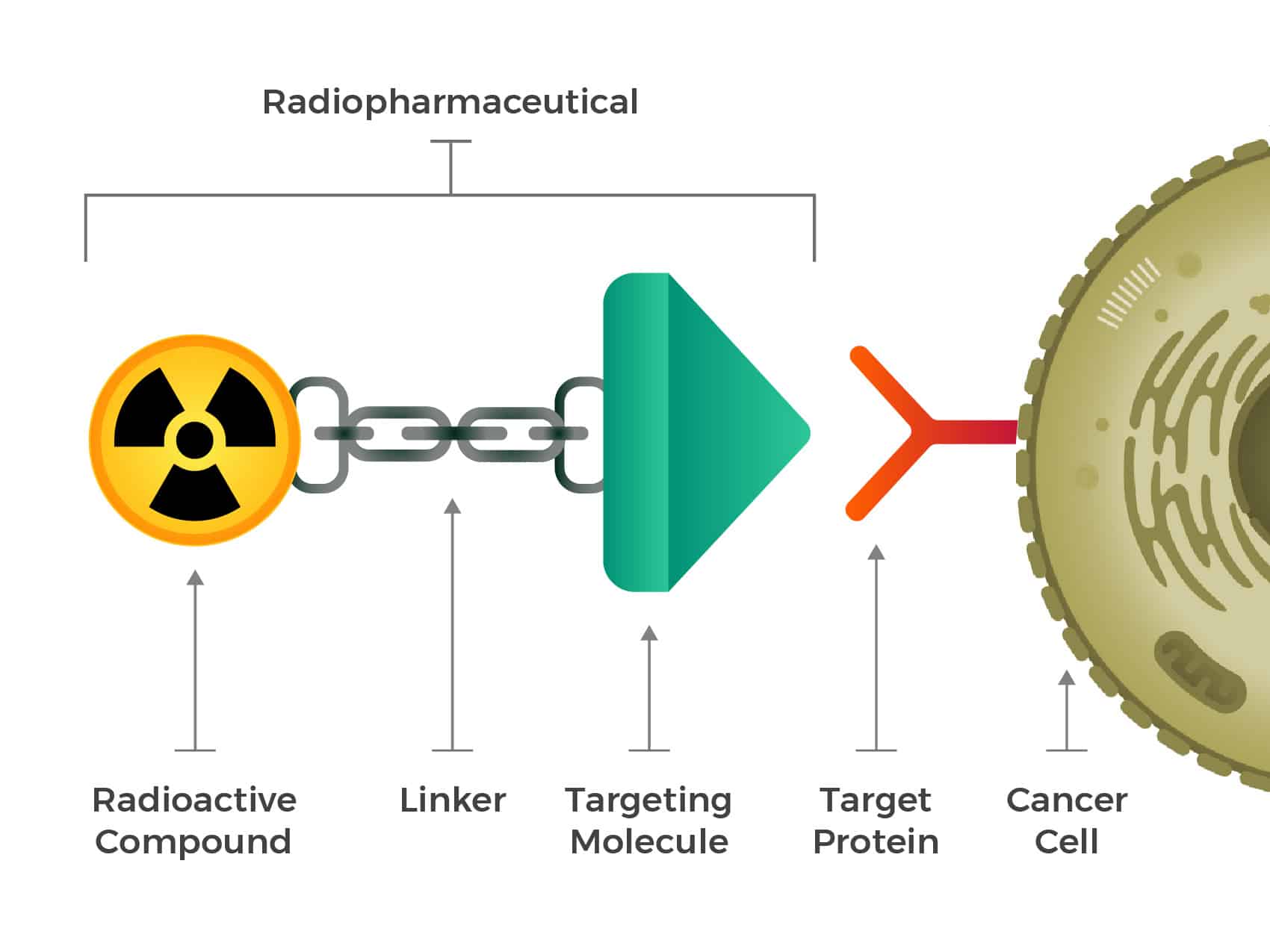
Radioactivity has been utilized as a treatment for cancer for over 100 years by delivering beams of radiation outside the body, causing widespread collateral damage. Radiopharmaceuticals represent the cutting edge of radionuclide-based radiation therapy. These are a class of drugs that deliver small amounts of radioactive isotopes directly to the target area, minimizing or eliminating the radiation exposure to surrounding tissue. These drugs can be used for both diagnosis and treatment [4]. Made up of a radioactive compound, a targeting molecule and a linker that joins the two, the promise of radiopharmaceuticals is precision: maximum damage where it is needed, minimal harm where it is not.
Radionuclide Targeted Alpha Therapy and Precision Cancer Treatment
A specific type of radiopharmaceutical used to treat cancer is radionuclide targeted alpha therapy (TAT). In these drugs, alpha-emitting radionuclides are prepared by chelation—effectively preventing toxic metal accumulation in healthy tissues—and linked to a target molecule, delivering the alpha radiation directly to the tumor [5]. The effectiveness of TAT lies in its name. Alpha particles are positively charged particles, consisting of two protons and two neutrons, and are about 8,000 times larger than beta particles. Alpha particles are emitted during the alpha decay of radionuclides, releasing enormous amounts of energy over a very short distance. This energy is cytotoxic, causing irreparable breaks in the DNA of cancer cells. In comparison, beta particles, as they are released, emit lower levels of damage over a higher range, making them less effective against cancer cells and more dangerous to healthy tissue [6]. Practically, this is a complex therapy with several complications. Suitable alpha-emitting isotopes are scarce and difficult to produce. Chelation is complex and unforgiving. Even when those hurdles are cleared, there remains the problem of radioactive daughter products, which can break free after decay and migrate to unintended parts of the body.
Why Pb-212 Is a Promising Radionuclide for Targeted Alpha Therapy
Among the most promising radionuclides for targeted alpha therapy is Lead-212 (Pb-212). Pb-212 is particularly prized because of its very short half-life, approximately 10.6 hours. This window of time is long enough for imaging and targeting the tumor, but not long enough to cause any serious damage to non-targeted regions. Pb-212 itself emits beta particles, but inside the body it acts as a generator for bismuth-212, an alpha-emitting radionuclide with a much shorter half-life of about 61 minutes [7]. This combination is unique, and it means that when timed correctly, the full effect of cytotoxic alpha radiation is delivered at the tumor site. Manufacture of Pb-212 is not straightforward, as it is most often produced in the decay chain of other radionuclides from which it must be isolated and purified. However, given its unique and promising position in radiopharmacology, there has been significant investment in increasing the supply of Pb-212.
Pb-212 Purification and the Challenge of Radionuclide Separation
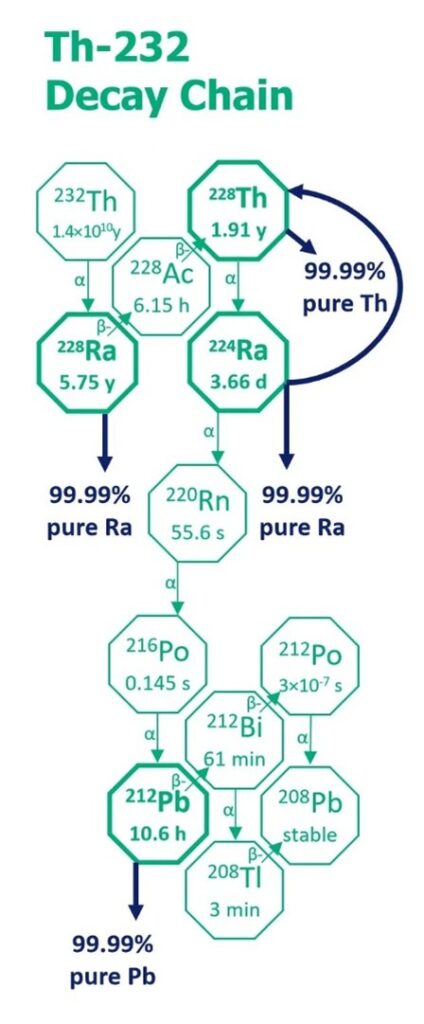
Pb-212 can be produced through the natural decay of thorium-232 or thorium-228, both of which generate a complex cascade of radioactive daughter isotopes. While this decay chain provides a viable source of Pb-212, it also creates a significant technical challenge. Pb-212 must undergo rapid radionuclide purification, separating it from chemically similar daughter products before it decays. For radiopharmaceutical applications, even trace impurities can compromise safety, regulatory compliance, and therapeutic performance.
Radionuclide Purification Using MRT™
IBC Advanced Technologies, Inc. (IBC Advanced Technologies - Molecular Recognition Technology™), through its subsidiary RadSep, Inc. (Radioisotope Separations Technology - RadSep), addresses this challenge using Molecular Recognition Technology™ (MRT™). Building on decades of experience in highly selective metal separations, IBC has developed MRT™ resins engineered specifically to recognize and bind Ra-228, Ra-224, Th-228 and Pb-212 with exceptional selectivity, resulting in radioisotopes of 99.99% chemical purity with greater than 99.9% recovery IBC launches RadSep, Inc.: Best-in-class, highly selective separations using Molecular Recognition Technology™ (MRT™) for production of critical radioisotopes essential to cancer therapies.
As shown in the diagram, Pb-212 can be produced from the decay of Th-232 or Th-228. The challenge is rapid Pb-212 purification from the other daughter isotopes in the decay chain. Highly selective MRT™ resins are used to produce Pb-212 at 99.99% purity and 99.9+% recovery. By selectively capturing Pb-212 as it forms and quickly releasing it in a simple, purified solution, MRT™-based systems support on-demand access to clinically relevant quantities of Pb-212. The speed of separation is critical. With a half-life of approximately 10.6 hours, delays directly reduce usable activity. MRT™ purification is designed to operate quickly and reproducibly, preserving yield while maintaining stringent quality requirements.
Equally important, MRT™ separations are inherently scalable and compatible with pharmaceutical manufacturing workflows. The solid-phase MRT™ resin format supports column-based operation, minimal reagent use, and straightforward integration into automated systems. Unlike less selective methods, MRT™ is very fast and compact. Excessive downstream processing and radioactive waste generation are minimized. Handling and disposal are greatly simplified and process economics are optimized.
Conclusion
Targeted alpha therapy promises a future where radiation damages tumors with extraordinary precision while sparing healthy tissue. Realizing that promise depends not only on biology and chemistry, but on reliable radionuclide purification technologies that can deliver high-purity alpha emitters at scale. Pb-212 occupies a uniquely powerful position in this landscape, but only if it can be produced, purified, and delivered on clinically meaningful timescales. By applying Molecular Recognition Technology™ to the purification of Pb-212, a practical approach that combines speed, selectivity, and scalability is enabled. In doing so, MRT™ removes one of the key bottlenecks limiting the broader adoption of next-generation radiopharmaceutical therapies.
Sources:
[1] Worldwide Cancer Research. How long has cancer been around? (2025 July). https://www.worldwidecancerresearch.org/cancer-and-research-information/myths-and-misinformation/how-long-has-cancer-been-around/
[2] Kucerova P, Cervinkova M (April 2016). "Spontaneous regression of tumour and the role of microbial infection--possibilities for cancer treatment". Anti-Cancer Drugs. 27 (4): 269–77. doi:10.1097/CAD.0000000000000337. PMC 4777220.
[3] National Cancer Institute. Radiation Therapy Side Effects. (May 2025) https://www.cancer.gov/about-cancer/treatment/types/radiation-therapy/side-effects
[4] Mayo Clinic. Radiopharmaceuticals. (2025 August) https://www.mayoclinic.org/tests-procedures/radiopharmaceutic/about/pac-20587480
[5] Mulford DA, Scheinberg DA, Jurcic JG. The promise of targeted {alpha}-particle therapy. J Nucl Med. 2005 Jan;46 Suppl 1:199S-204S. PMID: 15653670.
[6] Dekempeneer, Yana; Keyaerts, Marleen; Krasniqi, Ahmet; Puttemans, Janik; Muyldermans, Serge; Lahoutte, Tony; D’huyvetter, Matthias; Devoogdt, Nick (19 May 2016). "Targeted alpha therapy using short-lived alpha-particles and the promise of nanobodies as targeting vehicle". Expert Opinion on Biological Therapy. 16 (8): 1035–1047.
[7] Scaffidi-Muta JM, Abell AD. “Pb in targeted radionuclide therapy: a review.” EJNMMI Radiopharm Chem. 2025 Jul 1;10(1):34. doi: 10.1186/s41181-025-00362-7. PMID: 40591218; PMCID: PMC12214231.
