Scaling highly selective, high precision separations with Molecular Recognition Technology® (MRT™)
What began as a Nobel Prize-winning discovery in “host-guest” chemistry has evolved into a cornerstone of modern industrial separation. By engineering molecules that recognize and bind specific metals with atomic precision, Molecular Recognition Technology® (MRT™) is realizing the decades-old vision of rearranging atoms to create a new era of highly selective, green, and efficient industrial processes, from critical mineral recovery to life-saving radiopharmaceuticals and nuclear waste remediation.
Feynman’s Vision and Pedersen’s Discovery of Molecular Recognition [1]
Richard P. Feynman, 1965 Nobel Laureate in physics, made the following prophetic observation in 1960 about the prospect of rearranging atoms at the molecular level [2]: “What would the properties of materials be if we could really arrange the atoms the way we want them? They would be very interesting to investigate theoretically. I can’t see exactly what would happen, but I can hardly doubt that when we have some control of the arrangement of things on a small scale we will get an enormously greater range of possible properties that substances can have, and of different things that we can do.” How one molecule recognizes another has long intrigued scientists. The ability to explore this recognition property in a systematic way was accelerated by Pedersen’s discovery at DuPont in the 1960s that cyclic polyether hosts selectively complexed alkali metal ion guests [3]. This discovery laid the foundation for achievement of an elusive goal: creation of reagents of high specificity for a single chemical species with minimal interference from competing species. Two of the founders of IBC Advanced Technologies, Inc. (IBC), Dr. Reed M. Izatt and Dr. James J. Christensen, were among the first scientists, beginning in 1969, to quantitate the selectivity noted by Pedersen by measuring and compiling log K values for metal-macrocycle interactions [4]. An important result of Pedersen’s work was that it became immediately evident to the scientific community that well known organic chemistry synthetic procedures could be used to prepare an essentially unlimited variety of new ligands that could be used to investigate host-guest selectivity in a well-designed and selective manner for a wide range of species.
Professors Izatt and Christensen were the first non-DuPont scientists to visit Pedersen after publication of his cyclic polyether investigations and their early studies involved compounds provided by him. Pedersen had a great influence on Dr. Izatt and his future research direction in host-guest molecular recognition processes. Dr. Izatt’s vision of where this work might lead was given in his first publication describing his results [4a]: “there exist unusual opportunities for the synthesis of macrocyclic molecules that exhibit a high degree of selectivity in metal binding.” Pedersen’s contributions were truly monumental.
The Quantification of Selectivity: Remarkable Growth of Macrocyclic Chemistry Leading to the 1987 Nobel Prize in Chemistry
The message was out and, world-wide, scientists responded quickly and with fervor. A new field of science was born which early on was termed macrocyclic chemistry. A new term, ‘crown ether,’ became part of the chemistry vocabulary. This term reflected the similarity between a ‘crown’ on a monarch’s head and the fit of the cyclic polyether host to the metal guest. An illustration of this fit is shown in Figure 1 for the ligand 18-crown-6 containing K+ in its cavity.
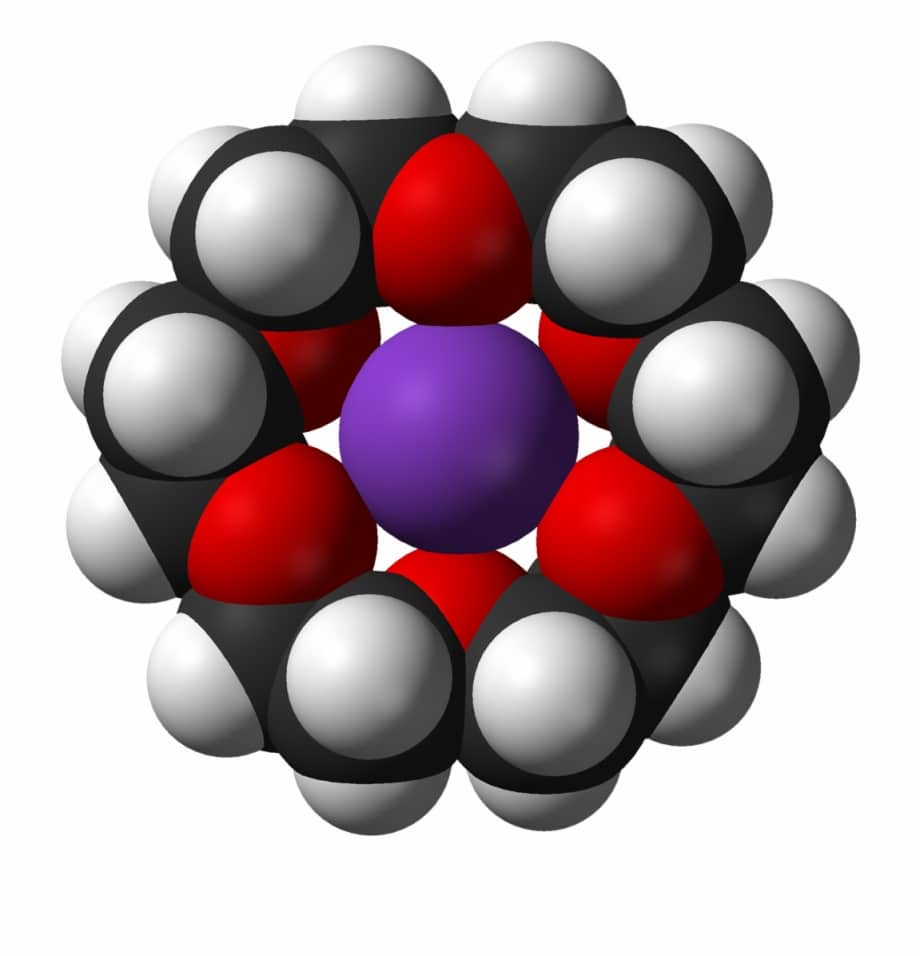 Figure 1. Depiction of a K+-18-crown-6 complex (18 carbon atoms, black, 6 oxygen binding atoms, red) showing the excellent fit of the K+(represented by the purple ball) in the 18-crown-6 cavity. The K+ is bound to the six oxygen atoms in the cavity. White balls represent hydrogen atoms. Source: [1]
Figure 1. Depiction of a K+-18-crown-6 complex (18 carbon atoms, black, 6 oxygen binding atoms, red) showing the excellent fit of the K+(represented by the purple ball) in the 18-crown-6 cavity. The K+ is bound to the six oxygen atoms in the cavity. White balls represent hydrogen atoms. Source: [1]
Ionic radii match for this host-guest pair, Na+ would be too small and ‘rattle around’ while Cs+ would be too large and lie above the cavity. The fit of the guest metal ions in the host cavity is consistent with log K values valid in methanol at 25 °C for 18-crown-6 interactions with these metal ions, i.e., Na+ = 4.36, K+ = 6.10, and Cs+ = 0.99 [5]. The specialties of Professors Izatt, Bradshaw and Christensen included the determination and interpretation of thermodynamic quantities (log K, ΔH, ΔS) for host-guest interactions. These quantities, especially log K, provide a quantitative basis for measuring and understanding molecular recognition processes. Over the years, IBC’s founders were involved in the preparation of numerous Chemical Reviews articles on thermodynamic quantities associated with macrocycle host-metal ion (and other) guest interactions [5]. The thermodynamic values in these compilations have been valuable because they provide a measure of molecular recognition and a basis for intelligent design of guest-selective hosts.
A decade after Pedersen’s landmark publication, the macrocyclic chemistry field had shown remarkable growth. Roeland Nolte remembers [6] that during a stay as visiting scientist in Donald Cram’s laboratory at UCLA in 1981, Cram told him that “after reading Pedersen’s paper he had become so excited that he had made the decision to completely change his research program.” This attitude was catching. Nolte goes on to say, “After having seen the potential of host-guest chemistry and the way it was approached by Cram, i.e., by designing compounds with the help of space-filling (CPK) models, we became fascinated and concluded that we should start a line of research in The Netherlands in which this new type of chemistry was incorporated.”
Another early worker, Jean-Marie Lehn, had coined the term, ‘supramolecular chemistry,’ to describe the broadening of the scope of host-guest chemistry which he and his research group spearheaded [7]. To quote Professor Lehn, “Beyond molecular chemistry, supramolecular chemistry aims at constructing highly complex, functional chemical systems from components held together by intermolecular forces.” These components can be visualized as host-guest systems bonded by intermolecular forces, which are much weaker than covalent chemical bonds. The guest systems may include organic guests as well as metal ions and the number and variety of hosts synthesized expanded far beyond macrocyclic compounds. Lehn’s article [7], particularly the early part, describes the development of supramolecular chemistry, which, in his case, resulted in his receipt of the 1987 Nobel Prize together with Donald Cram and Charles Pedersen. The 1987 Nobel Prize in Chemistry was awarded for the “development and use of molecules with structure-specific interactions of high selectivity.” [8]
Continued Research Leading to the 2016 Nobel Prize
The trajectory of the field continued with the 2016 Nobel Prize in Chemistry, awarded to Jean-Pierre Sauvage, Sir J. Fraser Stoddart, and Bernard Feringa for the design of molecular machines.[9] By developing molecules with controllable movements, these scientists realized Feynman’s suggestion that controlling the arrangement of things on a small scale would lead to an enormously greater range of possible properties and functions.
From Molecular Recognition to Industrial Selective Separations: The Birth of Molecular Recognition Technology® (MRT™) and its Impact on Global Metal Separations
During the 1970s and 1980s, the research efforts of Professors Izatt, Bradshaw and Christensen at Brigham Young University (BYU) were aimed at identifying the principles underlying molecular recognition processes in metal ion-organic ligand interactions. As these principles were learned and developed, their interests turned to studies of selective transport of metal ions in liquid membrane systems. Their goal was to study the ability of pre-designed hydrophobic macrocycles to selectively transport metal ions from one aqueous phase to a second aqueous phase separated by an organic liquid phase [10]. The hydrophobic metal-selective carrier macrocycle was located in the organic phase.
As their knowledge base grew, it occurred to Professors Izatt, Bradshaw and Christensen that a practical application of their metal selectivity studies might be achieved by attaching the macrocycle by a chemical bond to a solid substrate, such as silica gel, making regeneration and reuse of the valuable macrocycle possible. The achievement of this goal was reported in 1988 [11] and the same year IBC was formed as a spin-off company from their academic research program at BYU. The vision they had of the potential value of this new technology in metal separations and in metal recovery is seen in the opening remarks in the paper announcing this ground breaking achievement [11]:
“Sir: The recent permanent attachment of macrocycles such as the crown ethers to silica gel via a hydrocarbon-type linkage has made possible the design of systems capable of the selective and quantitative removal of cations from aqueous solutions. These systems can be operated indefinitely without loss of the expensive macrocycle and maintain the selectivity shown toward metal ions in aqueous solution by the particular macrocycle in the free state. These systems are of potential value in concentrating cations present at the nanogram-per-milliliter level, making their analysis by conventional procedures possible, and in selectively removing either wanted or unwanted metal cations from solutions in which they are present in the milligram-per-milliliter to nanogram-per-milliliter range. The latter application is treated in this correspondence using the alkaline-earth cations, Ag+, Hg2+, Tl+, and Pb2+.”
The term Molecular Recognition Technology® or MRT™ was coined in 1989 by Steven R. Izatt, President and CEO of IBC to describe the innovative technology and products of IBC, which were based on the pioneering work of Professors Izatt, Bradshaw and Christensen at BYU [12]. Adding the word “technology” to “molecular recognition” signifies the accomplishment of practical applications (i.e., products or systems for which a customer will pay money). MRT™ systems are engineered to perform selective separations in industry. Ligands are developed that not only have pre-determined selectivities but can be incorporated into commercial applications efficiently and cost-effectively.
Addressing Today’s Toughest Industrial Challenges
IBC has pioneered MRT™ applications across critical sectors where highly selective, high-precision separation is essential, including:
- Radiopharmaceuticals: In the burgeoning field of Targeted Alpha Therapy (TAT), MRT™ is utilized by IBC’s subsidiary, RadSep, to achieve 99.99% purity in isotopes like Actinium-225 and Lead-212.
- Green Energy & Lithium: Through GreenLit Pure Lithium®, IBC is revolutionizing the battery supply chain. The Direct Lithium to Product™ (DLP™) process enables the carbon-free production of superior battery-grade lithium directly from brine, ensuring a sustainable path for the global energy transition.
- Nuclear Waste Remediation: IBC’s manufacturing excellence is highlighted by the U.S. Department of Energy Secretary’s Award for scaling the production of MacroLig® 209, used to remediate legacy salt waste at the Savannah River Site.
- Critical Mineral Recovery: As global demand for green energy grows, MRT™ enables the highly selective recovery of Rare Earth Elements (REEs) and other critical minerals with a minimal environmental footprint.
Industrial Production: Scaling the Production of Macrocyclic Chemistry
Manufacturing macrocycles at an industrial scale requires specialized synthetic chemistry and advanced process engineering. With nearly four decades of experience, IBC manufactures macrocycles and chelating agents under strictly controlled conditions at industrial scale. This ensures consistent ligand structure, predictable binding behavior, and long-term durability in harsh industrial environments. IBC has a broad MacroLig® product line, including, for example, cryptands such as MacroLig® 403 (cryptand 2.2.1) and MacroLig® 404 (cryptand 2.2.2) that are used in radiopharmaceuticals, chemical synthesis and nuclear medicine; as well as calixarenes such as MacroLig® 209 (BOBCalixC6) used in nuclear waste remediation.
Award-Winning Manufacturing Excellence
IBC’s expertise in scaling macrocyclic chemistry was recognized by the U.S. Department of Energy Secretary’s Award. This honor was granted for IBC’s success in scaling the manufacturing of MacroLig® 209 (BOBCalixC6)—a critical component used in the remediation of legacy salt waste at the DOE’s Savannah River Site. This achievement demonstrates that MRT™ is not only a scientific breakthrough but a robust, industrial-scale solution.
The Advantages of the Macrocyclic Approach
- Extreme Selectivity: Distinguishes between ions with nearly identical chemical properties.
- High Stability: Resistant to chemical and radioactive degradation, essential for harsh nuclear and industrial environments.
- Green Chemistry: Operates under milder conditions, minimizing solvent use and secondary waste.
- Operational Efficiency: Enables high recovery efficiencies for valuable metals at concentrations as low as the nanogram-per-milliliter level.
Expanding Applications for Macrocyclic Chemistry
From its origins in the 1960s, macrocyclic chemistry has grown to achieve highly selective separations on an industrial scale that were previously impossible. The ongoing evolution of macrocyclic ligands remains a cornerstone for 21st-century innovation, offering transformative solutions for high-purity manufacturing, environmental remediation, green chemistry, and healthcare.
Put Nobel-Prize Precision to Work for Your Application
From critical mineral recovery to the production of high-purity medical isotopes, IBC’s Molecular Recognition Technology® (MRT™) systems are the global standard for highly selective separations.
- Explore MacroLig® Products – Our premier line of highly selective macrocycles and chelating agents designed for durability in industrial environments.
- Explore SuperLig® Applications – Industrial-scale SuperLig® resins engineered for large-volume metal recovery and environmental remediation.
- Discover AnaLig® Solutions – Precision AnaLig® resins designed for high-accuracy analytical applications and complex laboratory separations.
- Visit GreenLit Pure Lithium® – Learn about carbon-free Direct Lithium to Product® (DLP™) technology for production of superior battery-grade lithium.
- Discover RadSep – High-purity medical isotope solutions for Targeted Alpha Therapy (TAT) and cancer treatment.
- Contact Our Engineering Team – Discuss a custom MRT™ system tailored to your specific industrial requirements.
References
[1] Adapted from Izatt, R.M. 2022. “MRT™: A Disruptive Green Chemistry-based Highly Selective Metal Separations Process,” and “Molecular Recognition, Nobel Prizes and MRT™.”
[2] Feynman, R.P., 1960. There’s Plenty of Room at the Bottom, Engineering and Science, 23, 22-36.
[3] (a) Pedersen, C.J. 1967. Cyclic Polyethers and Their Complexes with Metal Salts, Journal of the American Chemical Society, 89, 7017-7036; (b) Pedersen, C.J. 1970. New Macrocyclic Polyethers, Journal of the American Chemical Society, 92, 391-394.
[4] (a) Izatt, R.M., Rytting, J.H., Nelson, D.P., Haymore, B.L., Christensen, J.J. 1969. Binding of Alkali Metal Ions by Cyclic Polyethers: Significance in Ion Transport Processes, Science, 164, 443-444; (b) Izatt, R.M., Nelson, D.P., Rytting, J.H., Haymore, B.L., Christensen, J.J., 1971. A Calorimetric Study of the Interaction in Aqueous Solution of Several Uni- and Bi-valent Metal Ions with the Cyclic Polyether Dicyclohexyl-18-Crown-6 at 10, 25, and 40o, Journal of the American Chemical Society, 93, 1619-1623; (c) Christensen, J.J., Hill, J.O., Izatt, R.M. 1971. Ion Binding by Synthetic Macrocyclic Compounds, Science, 174, 459-467.
[5] (a) Izatt, R.M., Pawlak, K., Bradshaw, J.S., Bruening, R.L. 1991. Thermodynamic and Kinetic Data for Macrocycle Interaction with Cations and Anions, Chemical Reviews, 91, 1721-2085; (b) Izatt, R.M., Bradshaw, J.S., Nielsen, S.A., Lamb, J.D., Christensen, J.J., Sen, D. 1985. Thermodynamic and Kinetic Data for Cation-Macrocycle Interaction, Chemical Reviews, 85, 271-339 (2,314 citations); (c) Christensen, J.J., Eatough, D.J., Izatt, R.M. 1974. The Synthesis and Ion Binding of Synthetic Multidentate Macrocyclic Compounds, Chemical Reviews, 74, 351-384 (1,037 citations).
[6] Nolte, R.J.M., Rowan, A.E., Elemans, J.A.A.W., Clipping Angel’s Wings, In Macrocyclic and Supramolecular Chemistry: How Izatt-Christensen Award Winners Shaped the Field, Izatt, R.M. (Ed), Wiley, Oxford, 2016, pp. 261-287.
[7] Lehn, J-M., From Supramolecular Chemistry towards Constitutional Dynamic Chemistry and Adaptive Chemistry, Chemical Society Reviews, 2007, 36, 151-160.
[8] The Nobel Prize in Chemistry 1987, http://www.nobelprize.org/nobel_prizes/chemistry/laureates/1987/ , Accessed December 23, 2021.
[9] Press Release: The Nobel Prize in Chemistry 2016 https://www.nobelprize.org/nobel_prizes/chemistry/laureates/2016/press.html Accessed December 27, 2021.
[10] (a) Izatt, R.M., Bradshaw, J.S., Lamb, J.D., Bruening, R.L., Emulsion and Supported Liquid Membranes, In Liquid Membranes: Chemical Applications; Araki, T., Tsukube, H., (Eds), CRC Press, Boca Raton, FL, 1990, Chapter 7.1, pp 123-140; (b) Izatt, R.M., Lamb, J.D., Bruening, R.L. 1988. Comparison of Bulk, Emulsion, Thin Sheet Supported, and Hollow Fiber Supported Liquid Membranes in Macrocycle-Mediated Cation Separations, Separation Science and Technology, 23, 1645-1658.
[11] Izatt, R.M., Bruening, R.L., Bruening, M.L., Tarbet, B.J., Krakowiak, K.E., Bradshaw, J.S., Christensen, J.J. 1988. Removal and Separation of Metal Ions from Aqueous Solutions Using a Silica-Gel-Bonded Macrocycle System, Analytical Chemistry, 60, 1825-1826.
[12] Izatt, N.E., Bruening, R.L., Krakowiak, K.E., Izatt, S.R. 2000. Contributions of Professor Reed M. Izatt to Molecular Recognition Technology: from Laboratory to Commercial Application, in Festschrift issue of Industrial & Engineering Chemistry Research, 39, 3405-3411.
